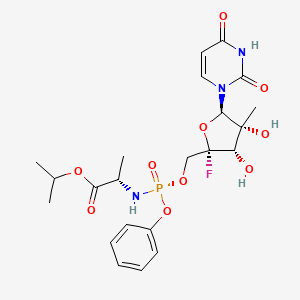
Golodirsen
- RNA, [P-deoxy-P-(dimethylamino)](2′,3′-dideoxy-2′,3′-imino-2′,3′-seco)(2’a→5′)(G-m5U-m5U-G-C-C-m5U-C-C-G-G-m5U-m5U-C-m5U-G-A-A-G-G-m5U-G-m5U-m5U-C), 5′-[P-[4-[[2-[2-(2-hydroxyethoxy)ethoxy]ethoxy]carbonyl]-1-piperazinyl]-N,N-dimethylphosphonamidate]
- Nucleic Acid Sequence
- Sequence Length: 25
| Formula |
C305H481N138O112P25
|
|---|---|
| CAS |
1422959-91-8
|
| Mol weight |
8647.2841
|
- Exon 53: NG-12-0163
- Golodirsen
- SRP 4053
Nucleic Acid Sequence
Sequence Length: 252 a 6 c 8 g 9 umodified
FDA APPROVED, Vyondys 53, 019/12/12
Antisense oligonucleotide
|
ゴロジルセン;
|
Duchenne muscular dystrophy (DMD variant amenable to exon 53 skipping)
VYONDYS 53 (golodirsen) injection is a sterile, aqueous, preservative-free, concentrated solution for dilution prior to intravenous administration. VYONDYS 53 is a clear to slightly opalescent, colorless liquid. VYONDYS 53 is supplied in single-dose vials containing 100 mg golodirsen (50 mg/mL). VYONDYS 53 is formulated as an isotonic phosphate buffered saline solution with an osmolality of 260 to 320 mOSM and a pH of 7.5. Each milliliter of VYONDYS 53 contains: 50 mg golodirsen; 0.2 mg potassium chloride; 0.2 mg potassium phosphate monobasic; 8 mg sodium chloride; and 1.14 mg sodium phosphate dibasic, anhydrous, in water for injection. The product may contain hydrochloric acid or sodium hydroxide to adjust pH.
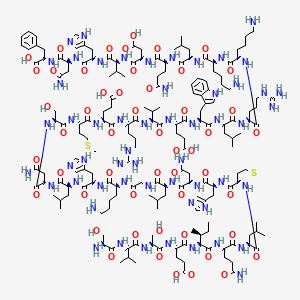
Golodirsen is an antisense oligonucleotide of the phosphorodiamidate morpholino oligomer (PMO) subclass. PMOs are synthetic molecules in which the five-membered ribofuranosyl rings found in natural DNA and RNA are replaced by a six-membered morpholino ring. Each morpholino ring is linked through an uncharged phosphorodiamidate moiety rather than the negatively charged phosphate linkage that is present in natural DNA and RNA. Each phosphorodiamidate morpholino subunit contains one of the heterocyclic bases found in DNA (adenine, cytosine, guanine, or thymine). Golodirsen contains 25 linked subunits. The sequence of bases from the 5′ end to 3′ end is GTTGCCTCCGGTTCTGAAGGTGTTC. The molecular formula of golodirsen is C305H481N138O112P25 and the molecular weight is 8647.28 daltons. The structure of golodirsen is:
 |
SIDE EFFECTS
- Hypersensitivity Reactions [see WARNINGS AND PRECAUTIONS]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In the VYONDYS 53 clinical development program, 58 patients received at least one intravenous dose of VYONDYS 53, ranging between 4 mg/kg (0.13 times the recommended dosage) and 30 mg/kg (the recommended dosage). All patients were male and had genetically confirmed Duchenne muscular dystrophy. Age at study entry was 6 to 13 years. Most (86%) patients were Caucasian.
VYONDYS 53 was studied in 2 double-blind, placebo-controlled studies.
In Study 1 Part 1, patients were randomized to receive once-weekly intravenous infusions of VYONDYS 53 (n=8) in four increasing dose levels from 4 mg/kg to 30 mg/kg or placebo (n=4), for at least 2 weeks at each level. All patients who participated in Study 1 Part 1 (n=12) were continued into Study 1 Part 2, an open-label extension, during which they received VYONDYS 53 at a dose of 30 mg/kg IV once weekly [see Clinical Studies].
In Study 2, patients received VYONDYS 53 (n=33) 30 mg/kg or placebo (n=17) IV once weekly for up to 96 weeks, after which all patients received VYONDYS 53 at a dose of 30 mg/kg.
Adverse reactions observed in at least 20% of treated patients in the placebo-controlled sections of Studies 1 and 2 are shown in Table 1.
Table 1: Adverse Reactions That Occurred in At Least 20% of VYONDYS 53-Treated Patients and at a Rate Greaterthan Placebo in Studies 1 and 2
| Adverse Reaction | VYONDYS 53 (N = 41) % |
Placebo (N = 21) % |
| Headache | 41 | 10 |
| Pyrexia | 41 | 14 |
| Fall | 29 | 19 |
| Abdominal pain | 27 | 10 |
| Nasopharyngitis | 27 | 14 |
| Cough | 27 | 19 |
| Vomiting | 27 | 19 |
| Nausea | 20 | 10 |
Other adverse reactions that occurred at a frequency greater than 5% of VYONDYS 53-treated patients and at a greater frequency than placebo were: administration site pain, back pain, pain, diarrhea, dizziness, ligament sprain, contusion, influenza, oropharyngeal pain, rhinitis, skin abrasion, ear infection, seasonal allergy, tachycardia, catheter site related reaction, constipation, and fracture.
Hypersensitivity reactions have occurred in patients treated with VYONDYS 53 [see WARNINGS AND PRECAUTIONS].
Antisense technology provides a means for modulating the expression of one or more specific gene products, including alternative splice products, and is uniquely useful in a number of therapeutic, diagnostic, and research applications. The principle behind antisense technology is that an antisense compound, e.g., an oligonucleotide, which hybridizes to a target nucleic acid, modulates gene expression activities such as transcription, splicing or translation through any one of a number of antisense mechanisms. The sequence specificity of antisense compounds makes them attractive as tools for target validation and gene functionalization, as well as therapeutics to selectively modulate the expression of genes involved in disease.
Duchenne muscular dystrophy (DMD) is caused by a defect in the expression of the protein dystrophin. The gene encoding the protein contains 79 exons spread out over more than 2 million nucleotides of DNA. Any exonic mutation that changes the reading frame of the exon, or introduces a stop codon, or is characterized by removal of an entire out of frame exon or exons, or duplications of one or more exons, has the potential to disrupt production of functional dystrophin, resulting in DMD.
Recent clinical trials testing the safety and efficacy of splice switching
oligonucleotides (SSOs) for the treatment of DMD are based on SSO technology to induce alternative splicing of pre-mRNAs by steric blockade of the spliceosome (Cirak et al., 2Q\ \; Goemans et al., 2011; Kinali et al., 2009; van Deutekom et al., 2007). However, despite these successes, the pharmacological options available for treating DMD are limited. Golodirsen is a phosphorodiamidate morpholino oligomer (PMO) designed to skip exon 53 of the human dystrophin gene in patients with DMD who are amendable to exon 53 skipping to restore the read frame and produce a functional shorter form of the dystrophin protein.
Although significant progress has been made in the field of antisense technology, there remains a need in the art for methods of preparing phosphorodiamidate morpholino oligomers with improved antisense or antigene performance.
PATENT
https://patents.google.com/patent/WO2017205880A1/en
Provided herein are processes for preparing phosphorodiamidate morpholino oligomers (PMOs). The synthetic processes described herein allow for a scaled-up PMO synthesis while maintaining overall yield and purity of a synthesized PMO.
Accordingly, in one aspect, provided herein is a process for preparing an oligomeric compound of Formula A):
(A).
In certain embodiments, provided herein is a process for preparing an oligomeric compound of Formula (G):
In yet another embodiment, the oligomeric compound of the disclosure including, for example, some embodiments of an oligomeric compound of Formula (G), is an oligomeric compound of Formula (XII):
(XII).
For clarity, the structural formulas including, for example, oligomeric compound of Formula (C) and Golodirsen depicted by Formula (XII), are a continuous structural formula from 5′ to 3′, and, for the convenience of depicting the entire formula in a compact form in the above structural formulas, Applicants have included various illustration breaks labeled “BREAK A” and “BREAK B.” As would be understood by the skilled artisan, for example, each indication of “BREAK A” shows a continuation of the illustration of the structural formula at these points. The skilled artisan understands that the same is true for each instance of “BREAK B” in the structural formulas above including Golodirsen. None of the illustration breaks, however, are intended to indicate, nor would the skilled artisan understand them to mean, an actual discontinuation of the structural formulas above including
Example 1: NCP2 Anchor Synthesis
1. Preparation of Meth l 4-Fluoro-3-Nitrobenzoate (1)
To a 100L flask was charged 12.7kg of 4-fluoro-3-nitrobenzoic acid was added 40kg of methanol and 2.82kg concentrated sulfuric acid. The mixture was stirred at reflux (65° C) for 36 hours. The reaction mixture was cooled to 0° C. Crystals formed at 38° C. The mixture was held at 0° C for 4 hrs then filtered under nitrogen. The 100L flask was washed and filter cake was washed with 10kg of methanol that had been cooled to 0° C. The solid filter cake was dried on the funnel for 1 hour, transferred to trays, and dried in a vacuum oven at room temperature to a constant weight of 13.695kg methyl 4-fluoro-3-nitrobenzoate (100% yield; HPLC 99%).
2. Preparation of 3-Nitro-4-(2-oxopropyl)benzoic Acid
A. (Z)-Methyl 4-(3 -Hydroxy- l-Methoxy-l-Oxobut-2-en-2-yl)-3-Nitrobenzoate (2)
To a 100L flask was charged 3.98kg of methyl 4-fluoro-3-nitrobenzoate (1) from the previous step 9.8kg DMF, 2.81kg methyl acetoacetate. The mixture was stirred and cooled to 0° C. To this was added 3.66kg DBU over about 4 hours while the temperature was maintained at or below 5° C. The mixture was stirred an additional 1 hour. To the reaction flask was added a solution of 8.15kg of citric acid in 37.5kg of purified water while the reaction temperature was maintained at or below 15° C. After the addition, the reaction mixture was stirred an addition 30 minutes then filtered under nitrogen. The wet filter cake was returned to the 100L flask along with 14.8kg of purified water. The slurry was stirred for 10 minutes then filtered. The wet cake was again returned to the 100L flask, slurried with 14.8kg of purified water for 10 minutes, and filtered to crude (Z)-methyl 4-(3 -hydroxy- 1 – methoxy-l-oxobut-2-en-2-yl)-3-nitrobenzoate.
B. 3-Nitro-4-(2-oxopropyl)benzoic Acid
2 3
The crude (Z)-m ethyl 4-(3 -hydroxy- 1-methoxy-l -ox obut-2-en-2-yl)-3-nitrobenzoate was charged to a 100L reaction flask under nitrogen. To this was added 14.2kg 1,4-dioxane and the stirred. To the mixture was added a solution of 16.655kg concentrated HC1 and 13.33kg purified water (6M HC1) over 2 hours while the temperature of the reaction mixture was maintained below 15° C. When the addition was complete, the reaction mixture was heated at reflux (80° C) for 24 hours, cooled to room temperature, and filtered under nitrogen. The solid filter cake was triturated with 14.8kg of purified water, filtered, triturated again with 14.8kg of purified water, and filtered. The solid was returned to the 100L flask with 39.9kg of DCM and refluxed with stirring for 1 hour. 1.5kg of purified water was added to dissolve the remaining solids. The bottom organic layer was split to a pre-warmed 72L flask, then returned to a clean dry 100L flask. The solution was cooled to 0° C, held for 1 hour, then filtered. The solid filter cake was washed twice each with a solution of 9.8kg DCM and 5kg heptane, then dried on the funnel. The solid was transferred to trays and dried to a constant weight of 1.855kg 3-Nitro-4-(2-oxopropyl)benzoic Acid. Overall yield 42% from compound 1. HPLC 99.45%.
3. Preparation of N-Tritylpiperazine Succinate (NTP)
To a 72L jacketed flask was charged under nitrogen 1.805kg triphenylmethyl chloride and 8.3kg of toluene (TPC solution). The mixture was stirred until the solids dissolved. To a 100L jacketed reaction flask was added under nitrogen 5.61kg piperazine, 19.9kg toluene, and 3.72kg methanol. The mixture was stirred and cooled to 0° C. To this was slowly added in portions the TPC solution over 4 hours while the reaction temperature was maintained at or below 10° C. The mixture was stirred for 1.5 hours at 10° C, then allowed to warm to 14° C. 32.6kg of purified water was charged to the 72L flask, then transferred to the 100L flask while the internal batch temperature was maintained at 20+/-50 C. The layers were allowed to split and the bottom aqueous layer was separated and stored. The organic layer was extracted three times with 32kg of purified water each, and the aqueous layers were separated and combined with the stored aqueous solution.
The remaining organic layer was cooled to 18° C and a solution of 847g of succinic acid in 10.87kg of purified water was added slowly in portions to the organic layer. The mixture was stirred for 1.75 hours at 20+/-50 C. The mixture was filtered, and the solids were washed with 2kg TBME and 2kg of acetone then dried on the funnel. The filter cake was triturated twice with 5.7kg each of acetone and filtered and washed with 1kg of acetone between triturations. The solid was dried on the funnel, then transferred to trays and dried in a vacuum oven at room temperature to a constant weight of 2.32kg of NTP. Yield 80%. 4. Preparation of (4-(2-Hydroxypropyl)-3-NitrophenyI)(4-Tritylpiperazin-l-yl)Methanone A. Preparation of l-(2-Nitro-4(4-Tritylpiperazine-l-Carbonyl)Phenyl)Propan-2-one
3 4
To a 100L jacketed flask was charged under nitrogen 2kg of 3-Nitro-4-(2- oxopropyl)benzoic Acid (3), 18.3 kg DCM, 1.845kg N-(3-dimethylaminopropyl)-N’- ethylcarbodiimide hydrochloride (EDC.HC1). The solution was stirred until a homogenous mixture was formed. 3.048kg of NTP was added over 30 minutes at room temperature and stirred for 8 hours. 5.44kg of purified water was added to the reaction mixture and stirred for 30 minutes. The layers were allowed to separate and the bottom organic layer containing the product was drained and stored. The aqueous layer was extracted twice with 5.65kg of DCM. The combined organic layers were washed with a solution of 1.08kg sodium chloride in 4.08kg purified water. The organic layers were dried over 1.068kg of sodium sulfate and filtered. The sodium sulfate was washed with 1.3kg of DCM. The combined organic layers were slurried with 252g of silica gel and filtered through a filter funnel containing a bed of 252g of silica gel. The silica gel bed was washed with 2kg of DCM. The combined organic layers were evaporated on a rotovap. 4.8kg of THF was added to the residue and then evaporated on the rotovap until 2.5 volumes of the crude l-(2-nitro-4(4-tritylpiperazine-l- carbonyl)phenyl)propan-2-one in THF was reached.
B. Preparation of (4-(2-Hydroxypropyl)-3-NitrophenyI)(4-Tritylpiperazin-l- yl)Methano
To a 100L jacketed flask was charged under nitrogen 3600g of 4 from the previous step and 9800g THF. The stirred solution was cooled to <5° C. The solution was diluted with 11525g ethanol and 194g of sodium borohydride was added over about 2 hours at <5° C. The reaction mixture was stirred an additional 2 hours at <5° C. The reaction was quenched with a solution of about 1.1kg ammonium chloride in about 3kg of water by slow addition to maintain the temperature at <10° C. The reaction mixture was stirred an additional 30 minutes, filtered to remove inorganics, and recharged to a 100L jacketed flask and extracted with 23kg of DCM. The organic layer was separated and the aqueous was twice more extracted with 4.7kg of DCM each. The combined organic layers were washed with a solution of about 800g of sodium chloride in about 3kg of water, then dried over 2.7kg of sodium sulfate. The suspension was filtered and the filter cake was washed with 2kg of DCM. The combined filtrates were concentrated to 2.0 volumes, diluted with about 360g of ethyl acetate, and evaporated. The crude product was loaded onto a silica gel column of 4kg of silica packed with DCM under nitrogen and eluted with 2.3kg ethyl acetate in 7.2kg of DCM. The combined fractions were evaporated and the residue was taken up in 11.7kg of toluene. The toluene solution was filtered and the filter cake was washed twice with 2kg of toluene each. The filter cake was dried to a constant weight of 2.275kg of compound 5 (46% yield from compound 3) HPLC 96.99%. 5. Preparation of 2,5-Dioxopyrrolidin-l-yl(l-(2-Nitro-4-(4-triphenylmethylpiperazine-l Carbon l)Phenyl)Propan-2-yl) Carbonate (NCP2 Anchor)
3 NCP2 Anchor
To a 100L jacketed flask was charged under nitrogen 4.3kg of compound 5 (weight adjusted based on residual toluene by 1H MR; all reagents here after were scaled accordingly) and 12.7kg pyridine. To this was charged 3.160 kg of DSC (78.91 weight % by 1H NMR) while the internal temperature was maintained at <35° C. The reaction mixture was aged for about 22 hours at ambience then filtered. The filter cake was washed with 200g of pyridine. In two batches each comprising ½ the filtrate volume, filtrate wash charged slowly to a 100L jacketed flask containing a solution of about 11kg of citric acid in about 50 kg of water and stirred for 30 minutes to allow for solid precipitation. The solid was collected with a filter funnel, washed twice with 4.3kg of water per wash, and dried on the filter funnel under vacuum.
The combined solids were charged to a 100L jacketed flask and dissolved in 28kg of DCM and washed with a solution of 900g of potassium carbonate in 4.3kg of water. After 1 hour, the layers were allowed to separate and the aqueous layer was removed. The organic layer was washed with 10kg of water, separated, and dried over 3.5kg of sodium sulfate. The DCM was filtered, evaporated, and dried under vacuum to 6.16kg of NCP2 Anchor (114% yield).
Example 2: Anchor Loaded Resin Synthesis
To a 75L solid phase synthesis reactor was charged about 52L of NMP and 2600g of aminomethyl polystyrene resin. The resin was stirred in the NMP to swell for about 2 hours then drained. The resin was washed twice with about 39L DCM per wash, then twice with 39L Neutralization Solution per wash, then twice with 39L of DCM per wash. The NCP2 Anchor Solution was slowly added to the stirring resin solution, stirred for 24 hours at room temperature, and drained. The resin was washed four times with 39L of NMP per wash, and six times with 39L of DCM per wash. The resin was treated and stirred with ½ the DEDC Capping Solution for 30 minutes, drained, and was treated and stirred with the 2nd ½ of the DEDC Capping Solution for 30 minutes and drained. The resin was washed six times with 39L of DCM per wash then dried in an oven to constant weight of 3573.71g of Anchor Loaded Resin.
Example 3: Preparation of Activated EG3 Tail
1. Preparation of Trityl Piperazine Phenyl Carbamate 35
To a cooled suspension of NTP in dichloromethane (6 mL/g NTP) was added a solution of potassium carbonate (3.2 eq) in water (4 mL/g potassium carbonate). To this two- phase mixture was slowly added a solution of phenyl chloroformate (1.03 eq) in
dichloromethane (2 g/g phenyl chloroformate). The reaction mixture was warmed to 20° C. Upon reaction completion (1-2 hr), the layers were separated. The organic layer was washed with water, and dried over anhydrous potassium carbonate. The product 35 was isolated by crystallization from acetonitrile. Yield=80%
2. Preparation of Carbamate Alcohol (36)
Sodium hydride (1.2 eq) was suspended in l-methyl-2-pyrrolidinone (32 mL/g sodium hydride). To this suspension were added triethylene glycol (10.0 eq) and compound 35 (1.0 eq). The resulting slurry was heated to 95° C. Upon reaction completion (1-2 hr), the mixture was cooled to 20° C. To this mixture was added 30% dichloromethane/methyl tert- butyl ether (v:v) and water. The product-containing organic layer was washed successively with aqueous NaOH, aqueous succinic acid, and saturated aqueous sodium chloride. The product 36 was isolated by crystallization from dichloromethane/methyl tert-butyl ether/heptane. Yield=90%.
3. Preparation of EG3 Tail Acid (37)
To a solution of compound 36 in tetrahydrofuran (7 mL/g 36) was added succinic anhydride (2.0 eq) and DMAP (0.5 eq). The mixture was heated to 50° C. Upon reaction completion (5 hr), the mixture was cooled to 20° C and adjusted to pH 8.5 with aqueous NaHC03. Methyl tert-butyl ether was added, and the product was extracted into the aqueous layer. Dichloromethane was added, and the mixture was adjusted to pH 3 with aqueous citric acid. The product-containing organic layer was washed with a mixture of pH=3 citrate buffer and saturated aqueous sodium chloride. This dichloromethane solution of 37 was used without isolation in the preparation of compound 38. 4. Preparation of Activated EG3 Tail (38)
To the solution of compound 37 was added N-hydroxy-5-norbornene-2,3-dicarboxylic acid imide (HONB) (1.02 eq), 4-dimethylaminopyridine (DMAP) (0.34 eq), and then l-(3- dimethylaminopropyl)-N’-ethylcarbodiimide hydrochloride (EDC) (1.1 eq). The mixture was heated to 55° C. Upon reaction completion (4-5 hr), the mixture was cooled to 20° C and washed successively with 1 : 1 0.2 M citric acid/brine and brine. The dichloromethane solution underwent solvent exchange to acetone and then to Ν,Ν-dimethylformamide, and the product was isolated by precipitation from acetone/N,N-dimethylformamide into saturated aqueous sodium chloride. The crude product was reslurried several times in water to remove residual Ν,Ν-dimethylformamide and salts. Yield=70% of Activated EG3 Tail 38 from compound 36.
Example 4: 50L Solid-phase Synthesis of
Golodirsen [Oligomeric Compound (XII)] Crude Drug Substance
1. Materials
Table 2: Starting Materials
Activated Phosphoramidochloridic acid, 1155373-31-1 C37H37CIN5O5P 698.2 C Subunit N,N-dimethyl-,[6-[4-
(benzoylamino)-2-oxo-l(2H)- pyrimidinyl]-4-
(triphenylmethyl)-2- morpholinyljmethyl ester
Activated Propanoic Acid, 2,2-dimethyl- 1155309-89-9 C5iH53ClN707P 942.2
DPG ,4-[[[9-[6-
Subunit [[[chloro(dimethylamino)phosp
hinyl]oxy]methyl]-4-
(triphenylmethyl)-2- morpholinyl]-2-[(2- phenylacetyl)amino]-9H-purin-
6-yl]oxy]methyl]phenyl ester
Activated Phosphoramidochloridic acid, 1155373-34-4 C3iH34ClN405P 609.1 T Subunit N,N-dimethyl-,[6-(3,4-dihydro- 5-methyl-2,4-dioxo- 1 (2H)- pyrimidinyl)]-4- (triphenylmethyl)-2- morpholinyljmethyl ester
Activated Butanedioic acid, 1- 1380600-06-5 C43H47N3Oio 765.9 EG3 Tail [3aR,4S,7R,7aS)-l,3,3a,4,7,7a- hexahydro- 1 ,3 -dioxo-4,7- methano-2H-isoindol-2-yl] 4- [2-[2-[2-[[[4-(triphenylmethyl)- 1- piperazinyl ] carb onyl ] oxy] ethox
y]ethoxy] ethyl] ester
Golodirsen.
Example 5: Purification of Golodirsen Crude Drug Substance
The deprotection solution from Example 4, part E, containing the Golodirsen crude drug substance was loaded onto a column of ToyoPearl Super-Q 650S anion exchange resin (Tosoh Bioscience) and eluted with a gradient of 0-35% B over 17 column volume (Buffer A: 10 mM sodium hydroxide; Buffer B: 1 M sodium chloride in 10 mM sodium hydroxide) and fractions of acceptable purity (CI 8 and SCX HPLC) were pooled to a purified drug product solution. HPLC: 93.571% (C18; Fig. 3) 88.270% (SCX; Fig. 4).
The purified drug substance solution was desalted and lyophilized to 1450.72 g purified Golodirsen drug substance. Yield 54.56 %; HPLC: 93.531% (Fig. 5; C18) 88.354% (Fig. 6; SCX).
PATENT
WO 2019067979
Duchenne Muscular Dystrophy (DMD) is a serious, progressively debilitating, and ultimately fatal inherited X-linked neuromuscular disease. DMD is caused by mutations in the dystrophin gene characterized by the absence, or near absence, of functional dystrophin protein that disrupt the mRNA reading frame, resulting in a lack of dystrophin, a critically important part of the protein complex that connects the cytoskeletal actin of a muscle fiber to the extracellular matrix. In the absence of dystrophin, patients with DMD follow a predictable disease course. Affected patients, typically boys, develop muscle weakness in the first few years of life, lose the ability to walk during childhood, and usually require respiratory support by their late teens. Loss of functional abilities leads to loss of independence and increasing caregiver burden. Once lost, these abilities cannot be recovered. Despite improvements in the standard of care, such as the use of glucocorticoids, DMD remains an ultimately fatal disease, with patients usually dying of respiratory or cardiac failure in their mid to late 20s.
Progressive loss of muscle tissue and function in DMD is caused by the absence or near absence of functional dystrophin; a protein that plays a vital role in the structure and function of muscle cells. A potential therapeutic approach to the treatment of DMD is suggested by Becker muscular dystrophy (BMD), a milder dystrophinopathy. Both dystrophinopathies are caused by mutations in the DMD gene. In DMD, mutations that disrupt the pre-mRNA reading frame,
referred to as “out-of-frame” mutations, prevent the production of functional dystrophin. In BMD, “in-frame” mutations do not disrupt the reading frame and result in the production of internally shortened, functional dystrophin protein.
An important approach for restoring these “out-of-frame” mutations is to utilize an antisense oligonucleotide to exclude or skip the molecular mutation of the DMD gene
(dystrophin gene). The DMD or dystrophin gene is one of the largest genes in the human body and consists of 79 exons. Antisense oligonucleotides (AONs) have been specifically designed to target specific regions of the pre-mRNA, typically exons to induce the skipping of a mutation of the DMD gene thereby restoring these out-of-frame mutations in-frame to enable the production of internally shortened, yet functional dystrophin protein.
The skipping of exon 53 in the dystrophin gene has been an area of interest for certain research groups due to it being the most prevalent set of mutations in this disease area, representing 8% of all DMD mutations. A prominent AON being developed by Sarepta
Therapeutics, Inc., for DMD patients that are amenable to exon 53 skipping is golodirsen.
Golodirsen is a phosphorodiamidate morpholino oligomer, or PMO. Another AON being developed by Nippon Shinyaku CO., LTD., for DMD patients that are amenable to exon 53 skipping is viltolarsen (NS-065 which is a PMO.
Exondys 51 ® (eteplirsen), is another PMO that was approved in 2016 by the United States Food and Drug Administration (FDA) for the treatment of Duchenne muscular dystrophy (DMD) in patients who have a confirmed mutation of the DMD gene that is amenable to exon 51 skipping. However, the current standard of care guidelines for the treatment of DMD in patients that are not amenable to exon 51 skipping include the administration of glucocorticoids in conjunction with palliative interventions. While glucocorticoids may delay the loss of ambulation, they do not sufficiently ameliorate symptoms, modify the underlying genetic defect or address the absence of functional dystrophin characteristic of DMD.
Previous studies have tested the efficacy of an antisense oligonucleotides (AON) for exon skipping to generate at least partially functional dystrophin in combination with a steroid for reducing inflammation in a DMD patient (see WO 2009/054725 and van Deutekom, et al., N. Engl. J. Med. 2007; 357:2677-86, the contents of which are hereby incorporated herein by reference for all purposes). However, treatment with steroids can result in serious complications, including compromise of the immune system, reduction in bone strength, and growth
suppression. Notably, none of the previous studies suggest administering an antisense
oligonucleotide for exon skipping with a non-steroidal anti-inflammatory compound to a patient for the treatment of DMD.
Thus, there remains a need for improved methods for treating muscular dystrophy, such as DMD and BMD in patients.
EXAMPLE 1
CAT- 1004 in Combination with M23D PMO Reduces Inflammation and Fibrosis in Mdx Mice.
To assess the effectiveness of a combination treatment of an exon skipping antisense oligonucleotide and an F-Kb inhibitor in Duchenne muscular dystrophy, M23D PMO and
CAT-1004 were utilized in the Mdx mouse model. The effect on inflammation and fibrosis was determined by analyzing samples of muscle taken from the quadriceps, of (1) wild-type mice treated with saline, (2) mdx mice treated with saline, (3) mdx mice treated with CAT-1004, (4) mdx mice treated with the M23D PMO, and (5) mdx mice treated with the M23D PMO in combination with CAT-1004. The tissue sections were analyzed for fibrosis by picrosirius red staining and for inflammation and fibrosis by Hematoxylin and Eosin (H&E) staining, as described in the Materials and Methods section above.
Treatment of Mdx mice with either M23D PMO or CAT-1004 as monotherapies resulted in a reduction of inflammation and fibrosis as compared to Mdx mice treated with saline.
Surprisingly, treatment of Mdx mice with the M23D PMO in combination with CAT-1004 resulted in reduced inflammation and fibrosis as compared with mice treated with CAT-1004
alone or M23D alone (Fig. 9). These results indicate the combination treatment enhances muscle fiber integrity.
EXAMPLE 2
Exon Skipping and Dystrophin Production in Mdx Mice Treated with the M23D
PMO and the M23D PMO in Combination with CAT- 1004
To analyze the extent of exon skipping and dystrophin production in mice treated with the M23D PMO in combination with CAT- 1004, samples of muscle were taken from the quadriceps, diaphragm, and heart of (1) wild-type mice treated with saline, (2) mdx mice treated with saline, (3) mdx mice treated with CAT- 1004, (4) mdx mice treated with the M23D PMO, and (5) mdx mice treated with the M23D PMO in combination with CAT- 1004. RT-PCR analysis for exon 23 skipping was performed as well as Western blot analysis to determine dystrophin protein levels.
Exon skipping was observed in the muscle of the quadriceps, diaphragm, and heart of the Mdx mice treated with the M23D PMO as well as mice treated with the M23D PMO in combination with CAT-1004 (Fig. 10). Surprisingly, enhanced dystrophin production was observed in the muscle of the quadriceps, diaphragm, and heart of the mice treated with the M23D PMO in combination with CAT-1004 as compared to treatment with M23D PMO monotherapy (Fig. 11). These results indicated the increase in dystrophin levels extended to the heart, a tissue known to have low efficiency of dystrophin upregulation by these agents when used alone. Notably, neither exon skipping nor dystrophin production were observed in mdx mice treated with CAT-1004 monotherapy (Figs. 10 and 11).
PATENT
WO 2019046755
PAPER
Methods in Molecular Biology (New York, NY, United States) (2018), 1828(Exon Skipping and Inclusion Therapies), 31-55.
PAPER
Human Molecular Genetics (2018), 27(R2), R163-R172.
///////////Golodirsen, ゴロジルセン , FDA 2019, ANTISENSE, Exon 53: NG-12-0163, SRP 4053, OLIGONUCLEOTIDE, Duchenne Muscular Dystrophy




















































![Graphical abstract Graphical abstract: One-pot synthesis of high molar activity 6-[18F]fluoro-l-DOPA by Cu-mediated fluorination of a BPin precursor](http://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9OB01758E&imageInfo.ImageIdentifier.Year=2019)